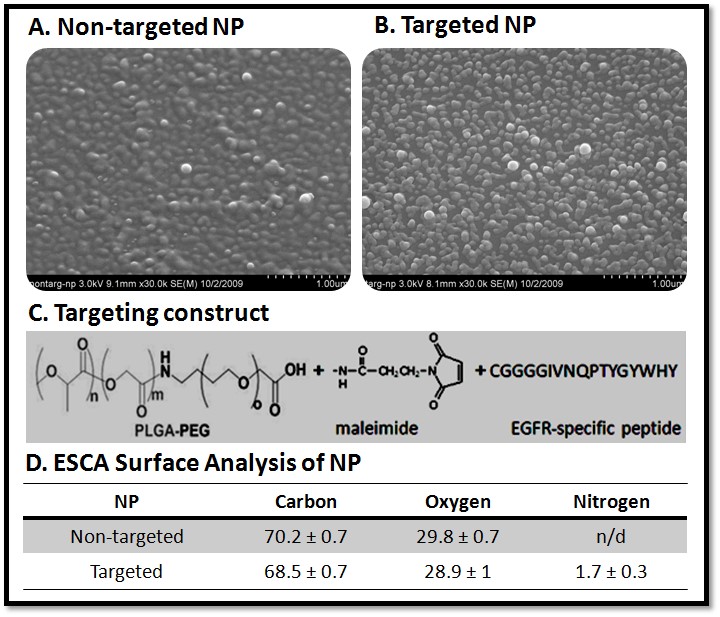
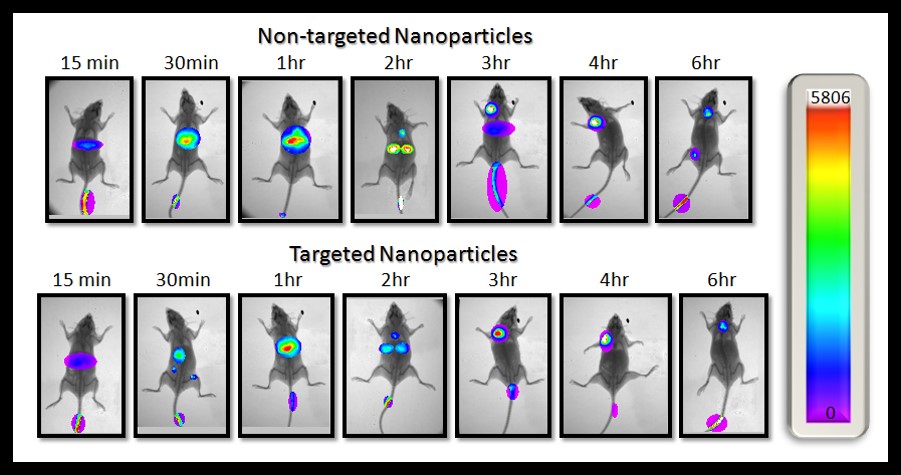
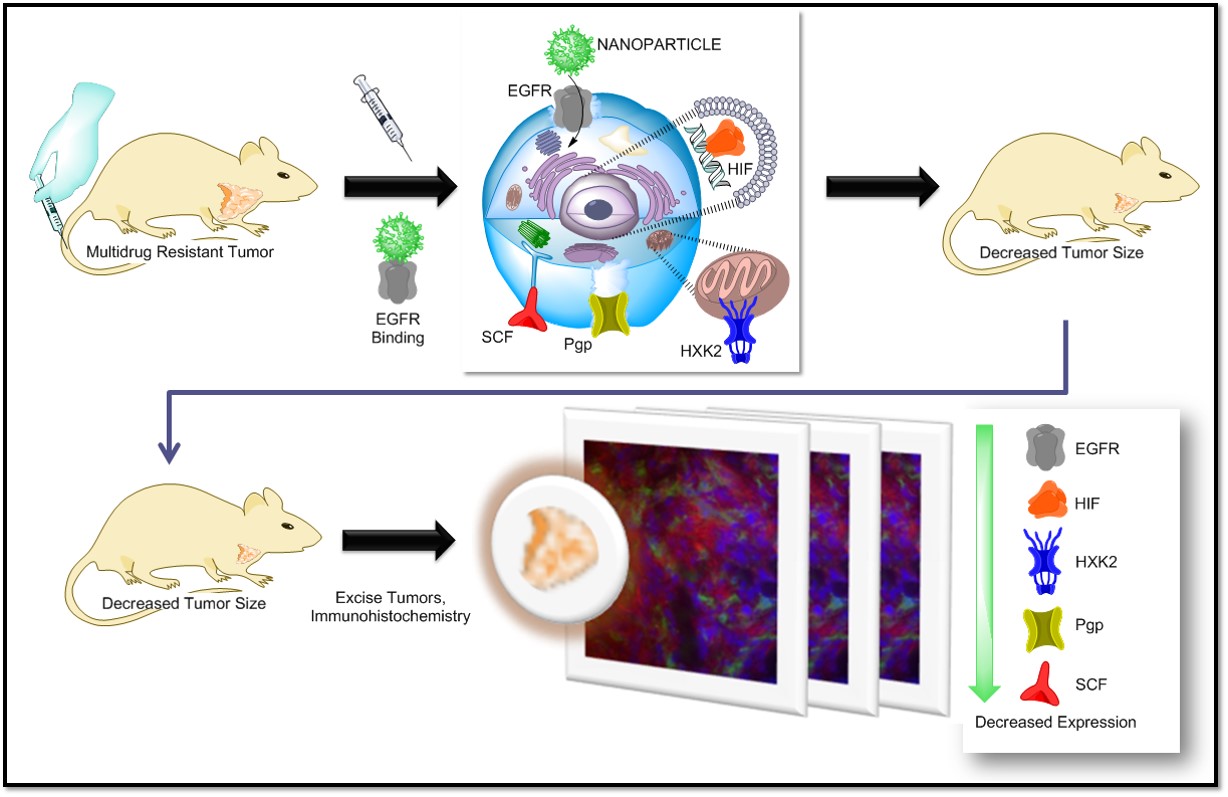
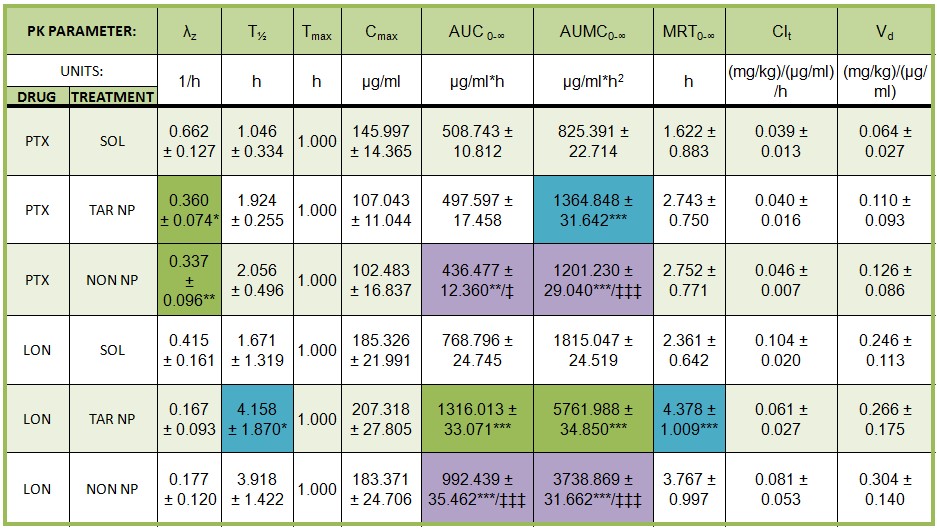
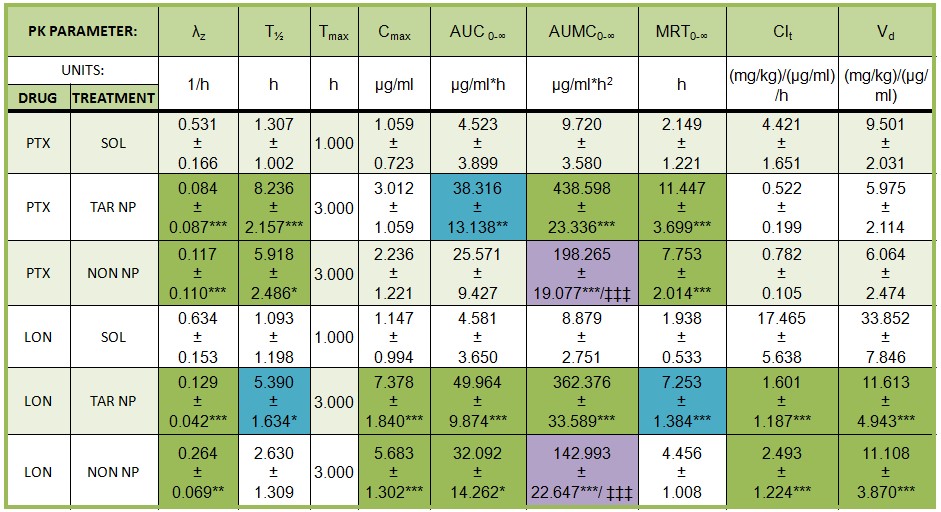
Current Projects
future Projects
Prior Studies
General Themes
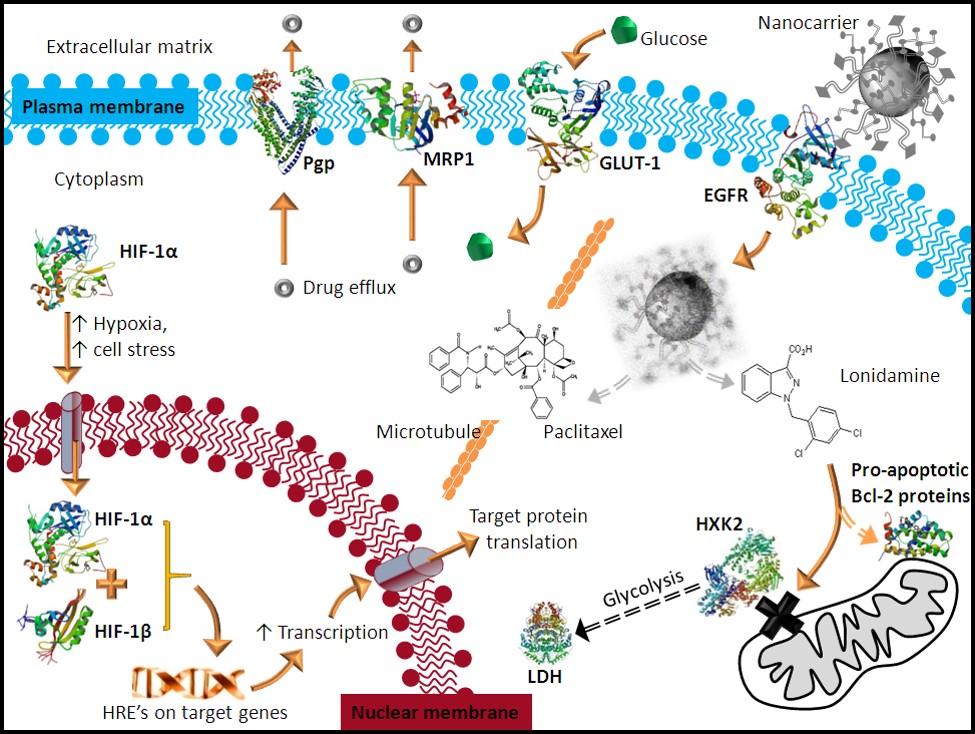
Exploiting Cancer Biology with Nanomedicine
Dr. Milane aims to develop molecularly targeted nanomedicines that exploit the hallmarks of cancer. The therapeutic design is to develop combination strategies that manipulate two or more survival strategies of multidrug resistant cancer in one nanomedicine formulation. For example, fragmenting the mitochondrial network while inducing the endoplasmic reticulum unfolded protein response.
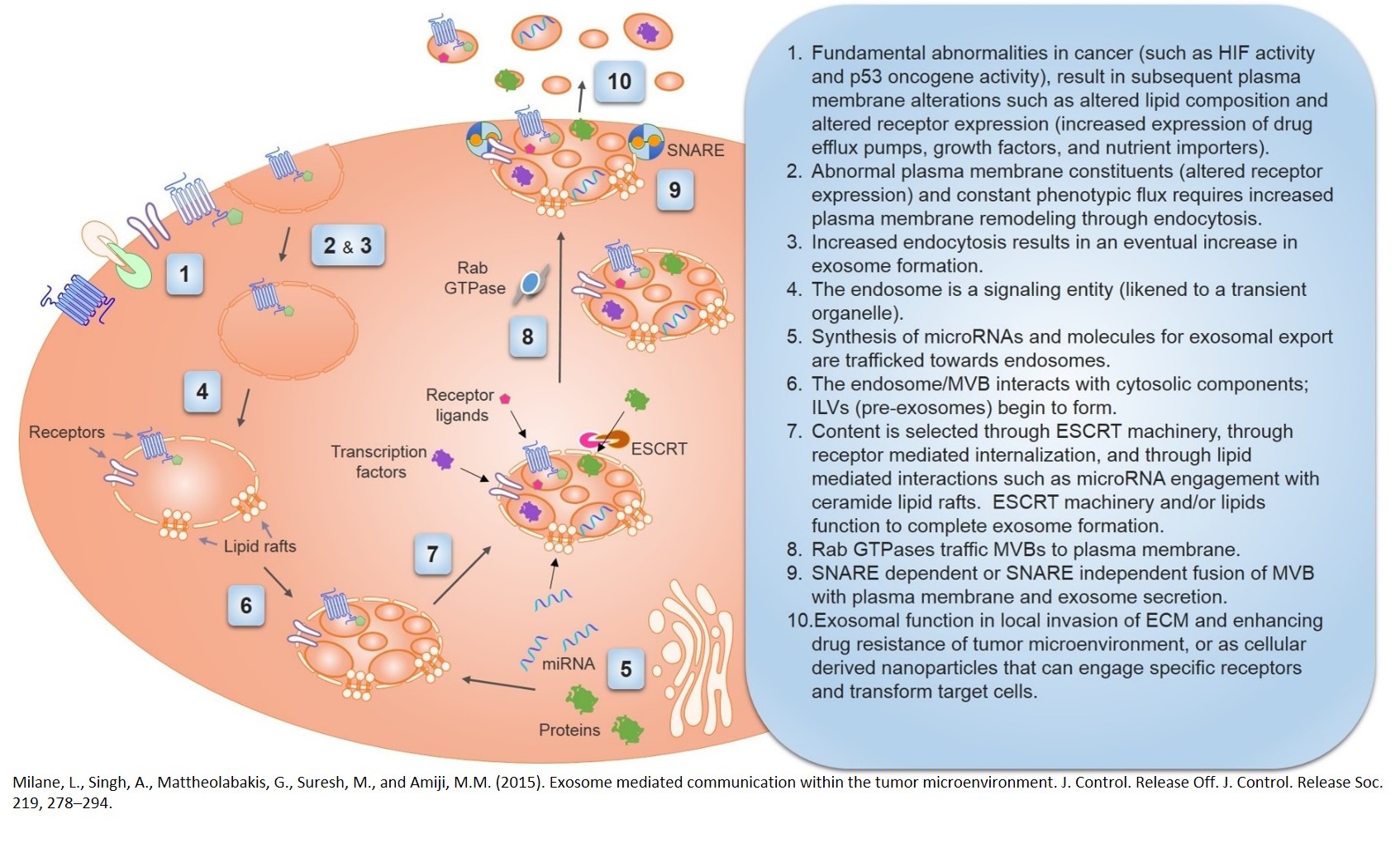
Cancer as Molecular Evolution
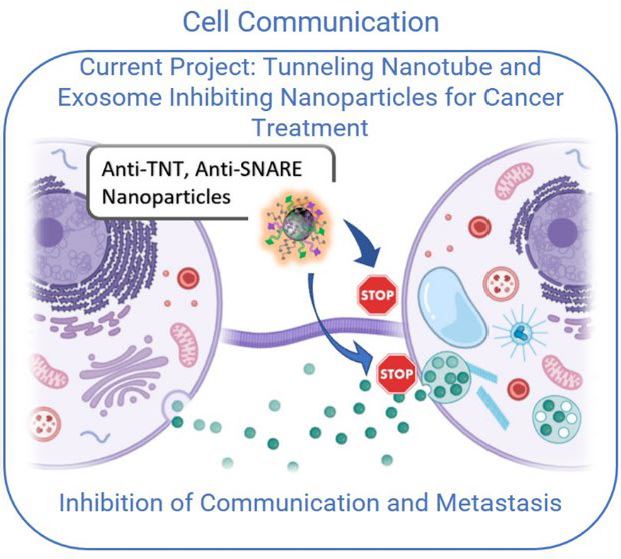
Dr. Milane has coined cancer as "survival of the fittest at its finest". Dr. Milane understands cancer in the context of molecular evolution. A cancer cell is a cell that adapts to selection pressures with the sole mission of cellular survival. Dr. Milane drew this image to illustrate cancer exosomal biology. Cancer exosomes aid survival as they function in metastasis, autocrine growth signaling, avoiding immune clearance, and macrophage polarization.
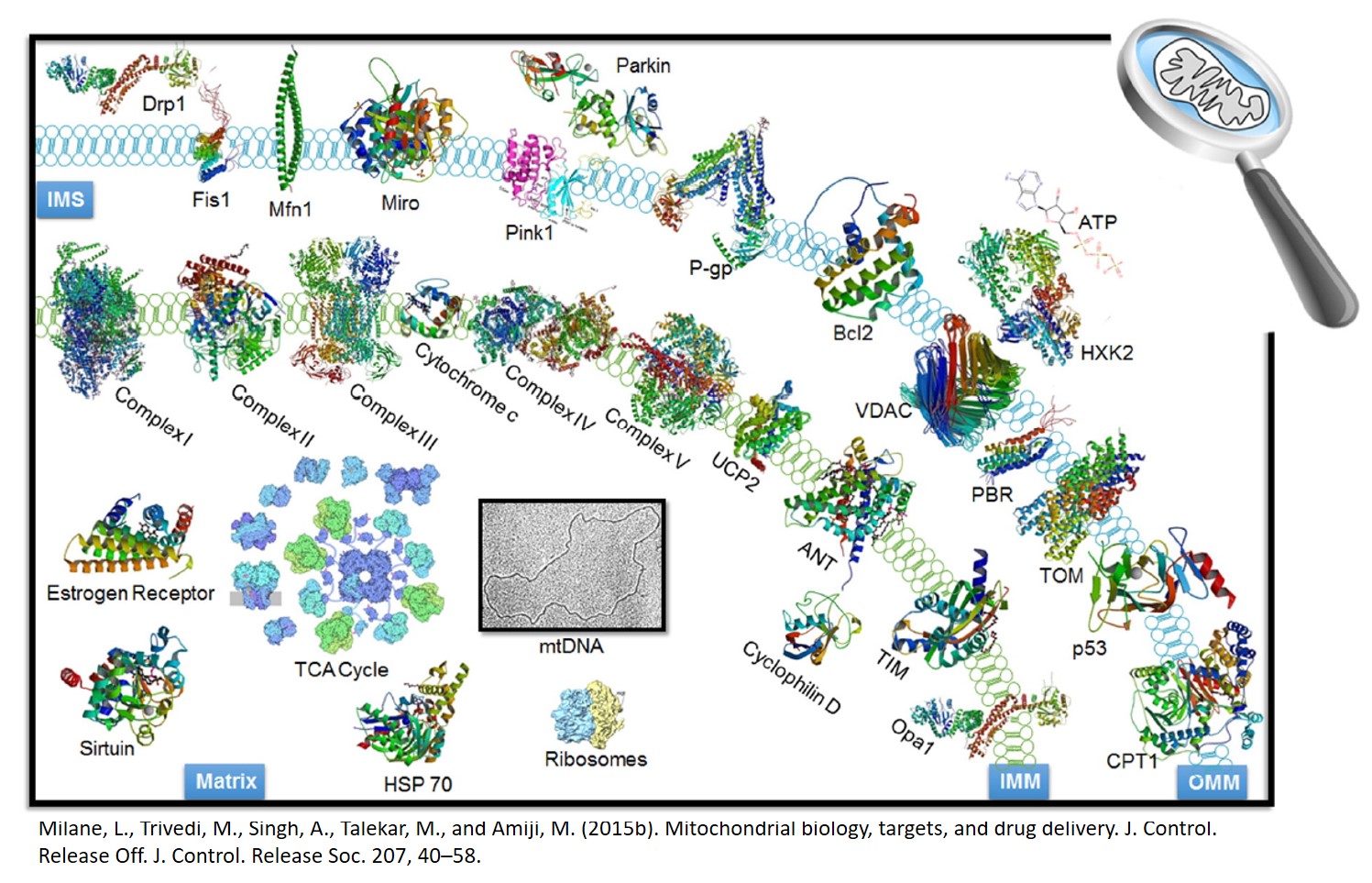
Mitochondrial Nanomedicine
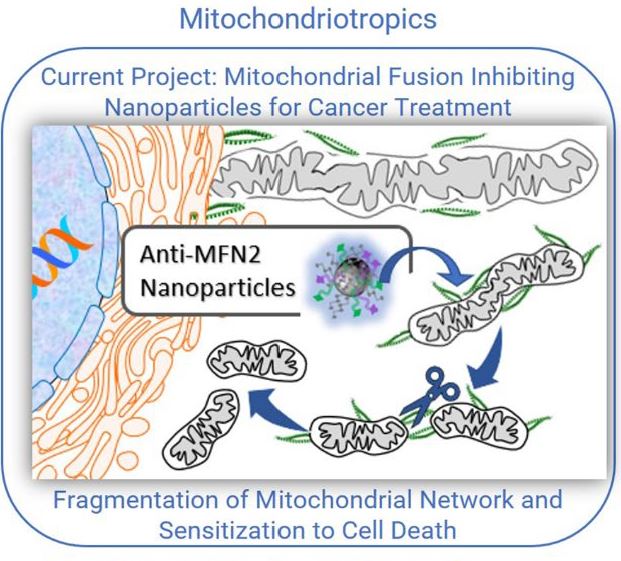
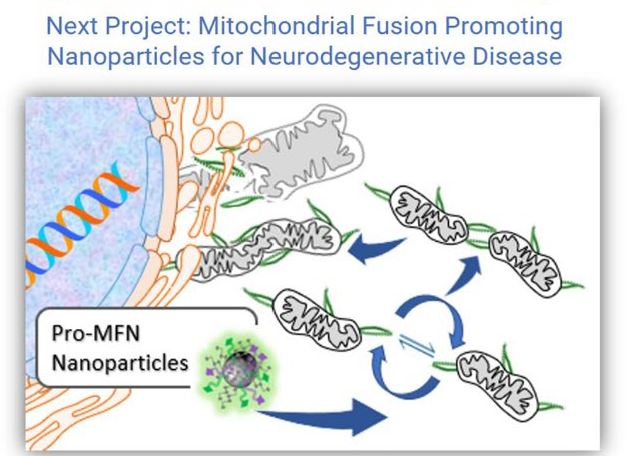
Many diseases beyond the classical mitochondrial myopathies are directly and indirectly associated with mitochondrial dysfunction. Mitochondrial dysfunction contributes to cancer, diabetes, the aging process, and neurodegenerative disease. For example, cancer is very much a mitochondrial disease as cancer cells are cells that are resistant to apoptosis, which is mediated by mitochondria. Cancer cells also revert to aerobic glycolysis (the Warburg Effect) for energy (less reliance on oxygen and mitochondrial oxidative phosphorylation and less toxic free radical production). Dr. Milane has identified the druggable targets in mitochondria and is interested developing mitochondriotropic drug delivery systems for multiple pathologies.
Connect
Dr. Milane is continually seeking to connect with other investigators and educators and enjoys productive research and teaching collaborations.